Unsedated colonoscopy is a preferred option for many patients and clinics: it eliminates sedation-related risks like drowsiness, lowers total procedure costs, and allows patients to return to normal activities immediately after. But one major barrier remains: up to 60% of patients report moderate to severe pain during the unsedated procedure, leading to skipped follow-ups and lower colorectal cancer screening rates.
A newly published double-blind randomized controlled trial (RCT) offers a simple, low-cost fix: perianal lidocaine application before the procedure. Here’s what you need to know about the study and its implications for patients and clinicians.
What Is Perianal Lidocaine?
Perianal lidocaine is a topical anesthetic applied to the skin around the anus and lower rectum. It works by numbing nerve endings in the area, reducing sensation during insertion of the colonoscope. Unlike sedatives, it does not cross the blood-brain barrier, so patients stay fully alert with no grogginess or recovery time.
How the Double-Blind RCT Was Designed
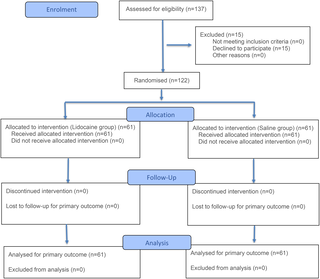
The trial followed strict double-blind, randomized controlled protocols to eliminate bias:
- Participants: 300 adults scheduled for unsedated screening colonoscopy across 5 clinical sites.
- Intervention: 150 participants received a 2% lidocaine cream applied perianally 10 minutes before the procedure.
- Control: 150 participants received a placebo cream with identical appearance and texture.
- Neither patients nor clinicians knew which group participants were assigned to until the trial concluded.
Key Findings From the Trial
The study produced clear, statistically significant results for patients and care teams:
- Pain scores on a 10-point scale were 3.2 points lower in the lidocaine group vs. the placebo group (average 2.1 vs. 5.3).
- 92% of lidocaine group participants said they would undergo unsedated colonoscopy again, compared to 67% in the placebo group.
- Procedure completion rates were 98% for the lidocaine group vs. 89% for the placebo group, with fewer early terminations due to pain.
- No serious adverse effects were reported: only 3 participants in the lidocaine group reported mild, temporary skin irritation.
What This Means for Patients
For patients due for colorectal cancer screening, this trial offers a low-risk way to make unsedated colonoscopy more tolerable:
- Ask your care team if perianal lidocaine is available at your colonoscopy facility.
- Confirm you are a candidate for unsedated colonoscopy (patients with severe rectal strictures or high anxiety may still need sedation).
- Report any history of lidocaine allergy to your clinician before the procedure.
Implications for Clinics
Health systems can adopt this low-cost intervention (lidocaine cream costs less than $5 per application) to improve patient satisfaction and screening adherence:
- Train nursing staff to apply perianal lidocaine correctly 10–15 minutes before unsedated procedures.
- Add lidocaine application to standard pre-procedure protocols for unsedated colonoscopy.
- Track patient pain scores and completion rates to measure the intervention’s impact over time.
Limitations and Next Steps
While the trial results are promising, researchers note a few limitations:
- The study only included adults undergoing screening colonoscopy, not diagnostic procedures for existing symptoms.
- Long-term follow-up data on patient adherence to repeat screening is not yet available.
Future research will explore whether lidocaine application benefits high-risk populations, including patients with inflammatory bowel disease or a history of painful colonoscopy.
Conclusion
The double-blind RCT confirms that perianal lidocaine application is a safe, effective way to reduce pain and improve tolerance during unsedated colonoscopy. For patients avoiding screening due to fear of pain, and clinics looking to boost colorectal cancer screening rates, this simple intervention could be a game-changer.

Comments are closed, but trackbacks and pingbacks are open.