For decades, scientists dismissed tRNA-derived fragments (tRFs) as mere cellular waste products. A new study turns that assumption on its head, revealing these small non-coding RNA molecules play a critical, previously unknown role in porcine fertility and a newly discovered cell death pathway called cuproptosis.
This research, focused on pig reproductive biology, uncovers a direct link between tRFs, granulosa-oocyte cell communication, and copper-driven cell death — with major implications for livestock breeding and human fertility research.
What Are tRNA-Derived Fragments?
tRNA-derived fragments (tRFs) are short RNA molecules, typically 14–30 nucleotides long, produced when full-length transfer RNAs (tRNAs) are cleaved under normal physiological conditions or cellular stress.
Once thought to be random degradation byproducts, tRFs are now recognized as functional regulators of gene expression. They bind to messenger RNA (mRNA) to block protein production, or interact with chromatin to alter gene activity. tRFs have been studied extensively in cancer and immune function, but their role in reproductive biology is only now emerging.
Porcine Granulosa-Oocyte Communication 101
In porcine ovaries, each immature oocyte is enclosed in a follicle surrounded by granulosa cells. These specialized cells form a bidirectional communication network with the oocyte: granulosa cells supply nutrients, hormones, and signaling molecules to support oocyte growth, while the oocyte secretes factors that regulate granulosa cell proliferation and function.
This crosstalk is essential for oocyte maturation, successful fertilization, and healthy fetal development. Disruptions to this communication are a leading cause of infertility, reduced litter sizes, and poor embryo quality in pigs.
The Novel Link to Cuproptosis
Cuproptosis is a recently discovered form of programmed cell death, first characterized in 2022. Unlike apoptosis or necrosis, cuproptosis is triggered by excessive accumulation of copper in cells, which binds to lipoylated proteins in the tricarboxylic acid (TCA) cycle, causing toxic protein aggregation and cell death.
While cuproptosis has been studied in cancer and neurodegenerative disease, its role in reproductive tissues has remained largely uncharacterized — until now.
How tRFs Tie It All Together
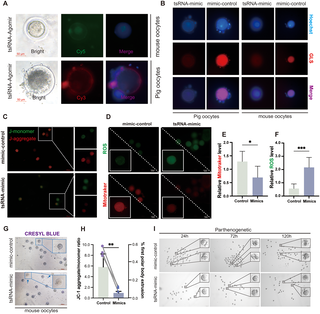
The new study mapped tRF expression in co-cultures of porcine granulosa and oocyte cells, compared to isolated cell populations. Researchers identified 12 specific tRFs that were consistently shuttled between the two cell types during normal communication.
Further experiments revealed these tRFs regulate key genes involved in copper homeostasis and cuproptosis in granulosa cells. When tRF levels were artificially reduced in co-cultures:
- Copper accumulation in granulosa cells increased by 40% within 24 hours
- Markers of cuproptosis (including FDX1 and LIAS protein aggregation) rose by 55%
- Oocyte maturation rates dropped by 32% due to disrupted granulosa cell support
- Supplementing cultures with the 12 identified tRFs reversed all these effects, restoring normal communication and reducing cuproptosis markers
Notably, oocyte-derived tRFs were found to directly target FDX1, a master regulator of the cuproptosis pathway, in granulosa cells — providing a direct molecular mechanism for this crosstalk.
Why This Matters for Industry and Research
For pork producers, improving sow fertility and litter sizes is a top priority for profitability. This research could lead to practical tools, including:
- Blood or follicular fluid biomarkers to identify high-fertility breeding sows earlier
- tRF-based supplements to improve oocyte quality in assisted reproductive technologies like in vitro fertilization (IVF) for pigs
- New strategies to reduce infertility caused by environmental or nutritional stress
For human fertility researchers, porcine reproductive biology shares striking similarities with human biology, making these findings highly translatable. The link between tRFs and cuproptosis also opens new avenues for studying infertility in humans, where cuproptosis has been linked to ovarian aging.
Conclusion
This study reshapes our understanding of tRNA-derived fragments, moving them from cellular waste to critical messengers in reproductive health. By linking tRFs to granulosa-oocyte communication and cuproptosis, researchers have uncovered a new regulatory axis that could transform both livestock breeding and human fertility research.
Future work will focus on testing tRF-based therapies in live animals, and exploring whether similar mechanisms operate in human ovaries — bringing us one step closer to solving complex fertility challenges.

Comments are closed, but trackbacks and pingbacks are open.