Introduction
Imagine a drug that knows exactly when and where to release its payload inside the brain, while simultaneously allowing doctors to see its journey in real‑time. This is no longer science fiction; stimuli‑responsive nanoplatforms are redefining how we treat central nervous system (CNS) disorders. In this article, we break down the technology, explore its therapeutic potential, and highlight the latest imaging breakthroughs.
What Are Stimuli‑Responsive Nanoplatforms?
Stimuli‑responsive nanoplatforms are engineered nanoparticles that alter their behavior in response to specific internal or external triggers. Common triggers include:
- pH changes: acidic microenvironments in tumors or inflamed tissue.
- Enzymes: overexpressed proteases or phosphatases in diseased brain regions.
- Temperature: mild hyperthermia applied via focused ultrasound.
- Redox conditions: elevated glutathione levels inside neurons.
- External fields: magnetic, light, or ultrasound cues.
When the trigger is encountered, the nanocarrier can:
- Release a therapeutic payload.
- Change surface charge to improve cellular uptake.
- Expose imaging agents for diagnostic contrast.
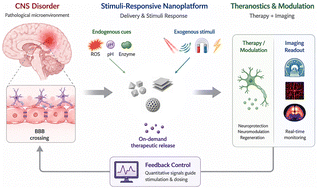
Why CNS Disorders Need Smart Delivery
The brain is protected by the blood‑brain barrier (BBB), which blocks >98% of small‑molecule drugs. Traditional delivery methods often result in:
- Low drug concentration at the target site.
- Systemic side effects.
- Inability to modulate disease progression dynamically.
Stimuli‑responsive nanoplatforms overcome these hurdles by:
- Crossing the BBB via receptor‑mediated transcytosis.
- Releasing drugs only after reaching the diseased microenvironment.
- Providing real‑time imaging feedback for dose adjustment.
Key Components of an Integrated Nanoplatform
1. Core Materials
Materials such as polymeric micelles, liposomes, mesoporous silica, and metal‑organic frameworks (MOFs) give structural stability and load capacity.
2. Targeting Ligands
Peptides (e.g., Angiopep‑2), antibodies, or aptamers bind to BBB transporters like transferrin receptor, ensuring efficient brain uptake.
3. Stimuli‑Sensitive Linkers
Acid‑labile hydrazone bonds, enzyme‑cleavable peptide sequences, or disulfide bridges trigger drug release under specific conditions.
4. Imaging Reporters
Gadolinium for MRI, near‑infrared dyes for optical imaging, or ^64Cu for PET enable non‑invasive tracking.
Therapeutic Applications
Neurodegenerative Diseases
For Alzheimer’s disease, nanocarriers can deliver anti‑amyloid antibodies that release in response to elevated β‑secretase activity, reducing off‑target exposure.
Brain Tumors
Glioblastoma exhibits a highly acidic microenvironment. pH‑responsive liposomes loaded with temozolomide release the drug only when pH < 6.8, sparing healthy tissue.
Stroke and Traumatic Brain Injury
Redox‑responsive nanoparticles unload antioxidants in regions with high reactive oxygen species (ROS), protecting neurons from oxidative damage.
Imaging Integration: Seeing the Nanoplatform in Action
Modern platforms combine therapy and diagnostics—so‑called “theranostics.” Examples include:
- MR‑visible silica nanocapsules: Gd‑chelated shells provide contrast while the core releases neuroprotective peptides.
- Photoacoustic nanoprobes: Near‑infrared absorbing particles enable ultrasound‑guided drug release monitoring.
- Dual‑modality PET/MRI agents: ^64Cu‑labeled polymers allow quantitative biodistribution analysis.
These imaging features help clinicians adjust dosing, verify BBB crossing, and assess treatment response early.
Challenges and Future Directions
- Safety and clearance: Long‑term accumulation of inorganic nanomaterials must be mitigated.
- Scale‑up manufacturing: Reproducible synthesis of complex nanostructures remains costly.
- Regulatory pathways: Combined therapeutic‑diagnostic products face multifaceted approval processes.
- Personalized triggers: Tailoring nanoplatforms to patient‑specific biomarkers will enhance efficacy.
Research is rapidly addressing these issues through biodegradable polymers, microfluidic production, and AI‑driven design of trigger sequences.
Conclusion
Stimuli‑responsive nanoplatforms bring a paradigm shift to CNS disorder treatment by uniting precise drug delivery, on‑demand release, and real‑time imaging. As safety profiles improve and manufacturing scales, we can expect these smart systems to move from bench to bedside, offering hope for conditions once deemed untreatable.

Comments are closed, but trackbacks and pingbacks are open.