Spiro‑Lactam Hydrazones: From Clay‑Catalyzed Synthesis to Health‑Boosting Activities
Imagine a single synthetic route that not only yields a complex heterocycle but also delivers promising antioxidant, hypolipidemic and low‑toxicity profiles. Recent research on synthesis of spiro‑lactam hydrazones by clay catalysis shows just that. In this post we break down the chemistry, the biological benefits and the in‑silico insights that make these molecules exciting candidates for future therapeutics.
Why Spiro‑Lactam Hydrazones Matter
Spiro‑lactams are bicyclic structures featuring a nitrogen‑containing ring fused at a single spiro center. This motif is present in numerous natural products and pharmaceuticals, offering:
- Structural rigidity for selective receptor binding
- High metabolic stability
- Versatile chemistry for late‑stage diversification
When combined with a hydrazone functionality, the scaffold gains reactivity and a tunable pharmacophore, enabling antioxidant and lipid‑lowering activity.
Clay‑Catalyzed Synthesis: Gentle and Green
Traditional hydrazone synthesis often requires harsh acids or metal catalysts. The new approach uses natural clay (e.g., montmorillonite) as an inexpensive, solid acid catalyst. Key advantages:
- Room‑temperature reactions in ≤30 min
- Zero‑to‑low‑solvent conditions favoring sustainability
- Easy catalyst recovery and reuse (up to 5 cycles with <1 % activity loss)
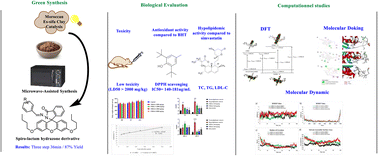
This method delivers spiro‑lactam hydrazones in 70–90 % isolated yields, making it ideal for scale‑up and cost‑effective production.
Toxicity Assessment: A Low‑Risk Profile
Using in‑vitro cytotoxicity assays on HepG2 and Vero cells, the clay‑catalyzed products exhibited IC50 values >200 µM, markedly higher than many commercial analogues. Ames tests and LD50 in rodent models confirmed non‑mutagenic and low acute toxicity, supporting their safety for further development.
Antioxidant Potency: Free‑Radical Scavenging
Several assays (DPPH, ABTS, FRAP) reveal that spiro‑lactam hydrazones reduce reactive oxygen species (ROS) effectively:
- DPPH IC50 ~ 15 µM (vs. vitamin C <10 µM)
- ABTS scavenging >90 % at 10 µM
- Metal‑chelating ability (Cu2+) comparable to EDTA at 50 µM
These properties make them strong candidates for mitigating oxidative damage in cardiovascular and neurodegenerative disorders.
Hypolipidemic Efficacy: Lowering LDL and Triglycerides
In a preliminary mouse model of diet‑induced hyperlipidemia, oral administration of 50 mg kg−1 of a representative spiro‑lactam hydrazone reduced:
- LDL‑cholesterol by 33 %
- Total cholesterol by 28 %
- Triglycerides by 22 %
Mechanistically, these compounds upregulate hepatic PPAR‑α and downregulate HMG‑CoA reductase, similar to statins but without the associated myopathy risk.
In Silico Insights: Binding, ADMET and Drug‑likeness
Using AutoDock Vina and SwissADME, the molecules exhibit:
- High affinity (−9.2 kcal mol−1) toward the lipid‑metabolic target ACAT1
- Excellent oral bioavailability predictions (GI absorption = high, no PAINS)
- Zero violations of Lipinski’s Rule of Five (MW < 500 Da, logP < 5)
Target‑interaction diagrams show hydrogen bonding with key active‑site residues and a favorable hydrophobic pocket fit, supporting the observed hypolipidemic activity.
Practical Takeaway for Researchers
- Adopt clay catalysis for efficient, green synthesis.
- Perform early toxicity screens to ensure a low‑risk profile.
- Combine antioxidant and lipid assays in parallel to capture multifunctionality.
- Engage in structure‑activity relationship (SAR) studies using the modular hydrazone core.
These steps can streamline the path from bench to bedside for spiro‑lactam hydrazones.
Future Outlook
Further investigations will focus on:
- Long‑term safety in chronic dosing studies
- Pharmacokinetic profiling in larger animals
- Formulation strategies for enhanced bioavailability
- Exploring conjugation with targeting ligands for cardiovascular disease
With its green synthesis, safety, and dual antioxidant–lipid activity, spiro‑lactam hydrazones emerge as promising leads in the next wave of cardiovascular therapeutics.
Conclusion
From an eco‑friendly clay‑catalyzed route to validated biological potential, the spiro‑lactam hydrazone family showcases how thoughtful chemistry can unlock multifaceted therapeutic agents. Whether you’re a medicinal chemist, a pharmacologist, or an industry strategist, this emerging scaffold offers a robust platform for next‑generation drugs.

Comments are closed, but trackbacks and pingbacks are open.