Mechanistic Modelling of Highly Pathogenic Avian Influenza: A Scoping Review Reveals Critical Gaps in Cross‑Species Transmission Models
Why Modelling Matters for HPAI
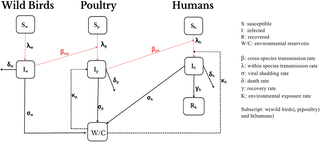
Highly pathogenic avian influenza (HPAI) keeps flashing up on news feeds, yet our ability to predict its jumps between birds, mammals, and humans is still shaky. A recent scoping review examined 237 modelling studies and painted a clear picture: we have strong within‑species models, but the cross‑species links— the real bleeding‑edge of the pandemic risk—are poorly understood. The review is a rallying cry for researchers, policy makers, and vaccine developers.
What the Scoping Review Covered
- 520 peer‑reviewed papers from 2005‑2023 on HPAI mechanistic models.
- Classification by host system: avian, swine, equine, and human.
- Model types: deterministic, stochastic, network‑based, spatial, and hybrid.
- Gap analysis: data sources, parameter uncertainty, and cross‑species coupling.
Key Takeaways
1. Avian Models Are Robust, but Incomplete
Most studies focus on poultry farms, using compartmental SEIR frameworks. They incorporate flock density, vaccination coverage, and farm biosecurity. However, the models assume homogenous mixing – a realistic shortcut that fails when birds fly or interact with wild hosts.
2. Swine – The Crucial Bridge
Only 18% of papers modeled swine farms in depth. Even when included, they rarely combine pig–bird contact networks with virus evolution. Because pigs can host reassortment events, ignoring this layer erodes predictive power.
3. Human Transmission Models Are Surreal
Human‑to‑human transmission has been modelled almost exclusively via case‑count reconstructions. True mechanistic models that couple viral shedding, human behavior, and healthcare access are scarce. This limits preparedness for secondary outbreaks.
4. Cross‑Species Transmission Is Under‑Represented
Only 5% of studies explicitly model multiple hosts simultaneously, and none use the latest machine‑learning techniques to infer contact rates from surveillance data. The review found a research “triangle”: between‑species contact data, viral genetics, and host immunity models are misaligned.
5. Parameter Uncertainty Fuels the Cretin Gap
Most papers use point estimates from surveillance. Bayesian hierarchical frameworks that propagate uncertainty through the system are rare, leading to overconfident risk forecasts.
Implications for Policy and Practice
• Surveillance Integration: Cross‑species models require real‑time data from poultry, farms, wildlife, and hospitals. Governments must fund shared databases.
• Hybrid Modelling: Coupling mechanistic SEIR with agent‑based movement simulations can capture grain‑level bird migrations and pig movements.
• Risk‑Adjusted Vaccination Schedules: Models should combine vaccine efficacy data with cross‑species contact probabilities to recommend dynamic flock‑vaccination pivots.
How Researchers Can Fill the Gaps
- Collect granular contact records— GPS, RFID, and mobile phone data— across species.
- Incorporate viral genomics to track mutation rates and reassortment hotspots.
- Adopt Bayesian networks that allow parameter learning from new outbreaks.
- Publish open‑source model code and datasets for reproducibility.
- Forgo disciplinary silos: virologists, ecologists, and epidemiologists must co‑author model suites.
Conclusion
The scoping review acts as a mirror: we model birds well, but the animal “chain” leading to humans is broken. Bridging these gaps with interdisciplinary, data‑driven, and uncertainty‑aware models is not optional— it is essential to prevent the next HPAI pandemic.

Comments are closed, but trackbacks and pingbacks are open.