For decades, the Suzuki-Miyaura cross-coupling has been the gold standard for forming carbon-carbon bonds in pharmaceutical, materials, and natural product synthesis. But its B-alkyl variant—used to couple alkylboranes with organic halides—has long frustrated chemists with poor yields, beta-hydride elimination side reactions, and limited substrate scope. Enter a game-changing alternative: palladium-catalyzed trans-vinylborane cross-coupling with alkyl bromides, a reversed-polarity approach that sidesteps these longstanding issues.
This method, which serves as a direct replacement for B-alkyl Suzuki reactions in C(sp2)-C(sp3) bond formation, leverages stereodefined trans-vinylboranes to deliver high yields, broad substrate tolerance, and perfect stereochemical control. Below, we break down how it works, why it outperforms traditional methods, and how you can start using it in your own lab.
What Makes This a Reversed-Polarity Alternative?
Traditional Suzuki-Miyaura reactions rely on a consistent polarity match: electrophilic organic halides are paired with nucleophilic boronic acids or boranes. In B-alkyl Suzuki reactions, this means coupling an electrophilic alkyl halide with a nucleophilic alkylborane. The problem? Alkyl-Pd intermediates formed during the reaction readily undergo beta-hydride elimination, breaking the very bond you are trying to form, while alkylboranes themselves are weak nucleophiles that slow the critical transmetallation step.
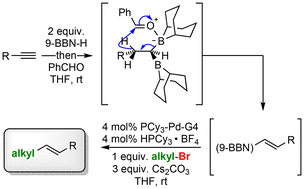
This trans-vinylborane cross-coupling flips that dynamic entirely. Instead of acting as a nucleophile, the trans-vinylborane serves as an electrophilic vinyl synthon—reversing the polarity of the borane partner relative to traditional B-alkyl Suzuki reactions. This avoids unstable alkyl-Pd intermediates entirely, as the vinyl-Pd species formed during the reaction have no beta-hydrogens to trigger elimination side reactions.
Key Components of the Reaction
Three core components drive this Pd-catalyzed cross-coupling, each optimized for reliability and broad scope:
1. Trans-Vinylboranes
These air-stable, stereodefined boranes feature a vinyl group bound to boron in the trans configuration. The trans geometry is non-negotiable: it ensures the vinyl group transfers cleanly to the Pd catalyst and retains its stereochemistry in the final product, delivering E-alkenes exclusively.
2. Alkyl Bromides
Unlike B-alkyl Suzuki reactions, which struggle with secondary or functionalized alkyl halides, this method works with primary, secondary, benzylic, and even alkyl bromides bearing sensitive functional groups like esters, ethers, and aryl halides. Alkyl iodides and chlorides can also be used with minor catalyst adjustments.
3. Palladium Catalyst and Ligands
Most protocols use pre-catalysts like Pd(dba)2 or Pd(OAc)2, reduced in situ to active Pd(0) species. Bulky, electron-rich phosphine ligands such as SPhos, XPhos, or RuPhos are critical to suppress side reactions and improve yields for sterically hindered substrates.
Advantages Over Traditional B-Alkyl Suzuki Reactions
This trans-vinylborane cross-coupling offers several concrete benefits for synthetic chemists:
- No beta-hydride elimination: Vinyl-Pd intermediates have no beta-hydrogens, eliminating the most common side reaction in B-alkyl Suzuki reactions.
- Broader substrate scope: Works with secondary alkyl bromides and functionalized substrates that fail in traditional B-alkyl Suzuki.
- Stereochemical fidelity: Trans-vinylboranes retain their geometry in the product, giving >99% E-selectivity for alkene products.
- Higher yields: Typical isolated yields range from 70-92% across most substrate classes, outperforming B-alkyl Suzuki by 20-40 percentage points for C(sp2)-C(sp3) couplings.
- Operational simplicity: Most reactions run at 60-80°C under inert atmosphere, with no need for strict anhydrous conditions beyond dry solvent.
Applications in Modern Synthesis
This reversed-polarity coupling has already found use across multiple fields of organic synthesis:
- Pharmaceutical research: Rapid assembly of vinyl-bearing alkyl side chains common in kinase inhibitors and antiviral agents.
- Materials science: Synthesis of conjugated polymers with tailored alkene linkages for organic solar cells and light-emitting diodes.
- Natural product synthesis: Streamlined construction of complex alkene-containing scaffolds, such as those found in polyunsaturated fatty acids and terpenes, without tedious protecting group strategies.
Getting Started in Your Lab
New to this method? Follow these beginner-friendly tips to optimize your first runs:
- Use freshly prepared or commercial trans-vinylboranes: While air-stable, they degrade slowly over 3-6 months of storage, so check expiration dates before use.
- Match ligand to substrate: Use SPhos for primary alkyl bromides, XPhos for secondary alkyl bromides, and RuPhos for sterically hindered or functionalized substrates.
- Avoid protic solvents: Use dry THF or toluene to prevent hydrolysis of the vinylborane, which would deactivate the catalyst.
- Screen reaction temperatures: Most substrates work at 60°C, but secondary alkyl bromides may require 80°C for full conversion.
Is This the Right Method for Your Project?
If you have struggled with low yields, side reactions, or limited substrate scope in B-alkyl Suzuki reactions, Pd-catalyzed trans-vinylborane cross-coupling offers a reliable, high-performing alternative. Its reversed-polarity design solves the core issues plaguing traditional alkyl-alkyl and alkyl-aryl Suzuki couplings, while delivering unmatched stereochemical control for alkene products.
Have you tried this method in your lab? Share your results in the comments below, or reach out for custom substrate optimization advice.

Comments are closed, but trackbacks and pingbacks are open.