Converting carbon dioxide into valuable chemicals is one of the most promising ways to tackle climate change while creating economic value. A recent breakthrough shows that a single‑atom copper catalyst (Cu₁/ZrO₂) paired with the zeolite SAPO‑34 can dramatically improve CO₂ hydrogenation to light olefins such as ethylene and propylene. In this article, we break down the science, why it matters, and how this technology could reshape the petrochemical industry.
Why Light Olefins from CO₂ Matter
Light olefins are the building blocks for plastics, synthetic fibers, and fuels. Traditionally, they are produced from petroleum‑based processes that emit large amounts of CO₂. By using captured CO₂ as the carbon source, the carbon footprint can be cut dramatically, turning a greenhouse gas into a feedstock.
The Catalyst Duo: Single‑Atom Cu₁/ZrO₂ + SAPO‑34
Two components work together synergistically:
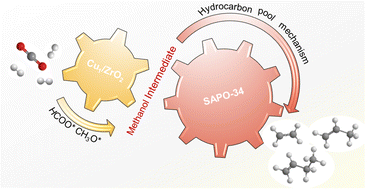
- Cu₁/ZrO₂ – Copper atoms are isolated on a zirconia support, offering high dispersion and unique electronic properties. This site efficiently activates CO₂ and H₂, forming surface-bound intermediates.
- SAPO‑34 – A small‑pore silicoaluminophosphate zeolite that selectively converts the intermediates into light olefins while suppressing unwanted side reactions such as methane formation.
The combination creates a “push‑pull” mechanism: Cu₁/ZrO₂ generates carbonyl‑rich species, and SAPO‑34 quickly extracts them as olefins, preventing over‑hydrogenation.
Key Performance Highlights
- Olefin selectivity: Up to 68 % of total carbon conversion ends as C₂–C₄ olefins.
- CO₂ conversion: 25–30 % at 350 °C and 3 MPa, surpassing conventional Cu/ZnO/Al₂O₃ systems.
- Stability: Minimal loss of activity over 200 h on stream, thanks to the strong metal‑support interaction that prevents copper sintering.
How the Reaction Works – A Simple Step‑by‑Step
- CO₂ activation: Single Cu atoms bind CO₂, bending the molecule and lowering the activation barrier for hydrogenation.
- Hydrogen addition: H₂ dissociates on adjacent Zr sites, delivering hydrogen atoms to the bound CO₂ to form formate and methoxy intermediates.
- Intermediate migration: These intermediates diffuse to the pores of SAPO‑34.
- Olefins formation: Inside SAPO‑34, a concerted dehydration‑dehydrogenation sequence releases C₂–C₄ olefins.
- Product desorption: Olefins exit the zeolite pores, while water and CO are removed with the gas stream.
Benefits for Industry and Environment
Adopting this catalyst system offers several advantages:
- Lower energy demand: The reaction proceeds at moderate temperatures (300‑350 °C), reducing furnace costs.
- Reduced greenhouse emissions: Direct utilization of CO₂ cuts net carbon output of olefin plants.
- Scalable design: Both Cu₁/ZrO₂ and SAPO‑34 are compatible with existing fixed‑bed reactors.
- Economic incentive: Producing high‑value olefins from waste CO₂ can generate new revenue streams for carbon‑capture facilities.
Challenges and Future Directions
While promising, the technology faces hurdles:
- Ensuring uniform single‑atom distribution on a large scale.
- Optimizing the zeolite pore structure to balance olefin selectivity and catalyst lifespan.
- Integrating the process with renewable hydrogen supply to achieve truly carbon‑negative production.
Research is already exploring alloyed single‑atom sites (e.g., Cu‑Ni) and hybrid zeolites to push selectivity beyond 80 %.
Takeaway
The marriage of single‑atom Cu₁/ZrO₂ and SAPO‑34 marks a pivotal step toward sustainable petrochemistry. By turning CO₂ into light olefins efficiently, this catalyst system offers a clear pathway to greener plastics and fuels, aligning economic goals with climate action.
Quick Reference
Keywords: CO₂ hydrogenation, single‑atom catalyst, Cu₁/ZrO₂, SAPO‑34, light olefins, sustainable petrochemistry

Comments are closed, but trackbacks and pingbacks are open.