Introduction
Industrial wastewater is loaded with synthetic dyes that resist conventional treatment. Eriochrome Black T (EBT) is a popular metal‑complexing dye used in textile and analytical labs, but its persistence poses serious environmental risks. Researchers have recently discovered a sunlight‑driven photocatalyst that can break down EBT within minutes: a highly efficient lanthanum‑doped silver phosphate (La‑Ag₃PO₄) nanocomposite decorated with zinc sulfide quantum dots (ZnS QDs). This post explains how the material works, why it outperforms traditional photocatalysts, and what it means for sustainable wastewater treatment.
Why Photocatalysis Is a Game‑Changer
Photocatalysis uses light energy to generate reactive oxygen species (ROS) that oxidize organic pollutants. The key advantages are:
- Solar compatibility: Sunlight provides a free, abundant energy source.
- Mild conditions: No extreme pH or temperature is required.
- Complete mineralization: Pollutants are converted to CO₂, H₂O, and inorganic ions.
For EBT, achieving rapid degradation under natural sunlight has been a major hurdle because most photocatalysts either absorb only UV light or suffer from fast electron–hole recombination.
Design of the La‑Doped Ag₃PO₄/ZnS Photocatalyst
1. Base material – Ag₃PO₄
Silver phosphate is a visible‑light active semiconductor with a narrow band gap (~2.4 eV). However, pure Ag₃PO₄ suffers from:
- Photo‑corrosion to metallic Ag.
- Rapid recombination of photogenerated charge carriers.
2. Role of Lanthanum (La) Doping
Inserting La³⁺ ions into the Ag₃PO₄ lattice creates oxygen vacancies and introduces mid‑gap states. This results in:
- Enhanced charge separation.
- Improved chemical stability against reduction.
- Shifted absorption edge toward longer wavelengths.
3. ZnS Quantum Dots (QDs) Decoration
ZnS QDs have a wide band gap (~3.6 eV) and a high conduction band (CB) potential. When anchored on La‑Ag₃PO₄, they act as:
- Electron sinks that accept electrons from the Ag₃PO₄ CB, suppressing recombination.
- Sensitizers that extend light harvesting into the UV‑visible region.
- Sites for the generation of •OH and ·O₂⁻ radicals.
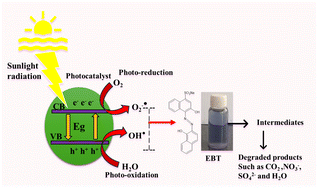
How the Catalyst Degrades Eriochrome Black T
The degradation pathway follows a classic photocatalytic mechanism:
- Light absorption: Sunlight excites electrons from the valence band (VB) of La‑Ag₃PO₄ to its CB, leaving holes in the VB.
- Charge transfer: Electrons are transferred to ZnS QDs, while La‑induced vacancies trap holes, extending their lifetime.
- Reactive species formation: Trapped electrons reduce O₂ to superoxide radicals (·O₂⁻); holes oxidize H₂O to hydroxyl radicals (•OH).
- Pollutant attack: •OH and ·O₂⁻ rapidly oxidize the azo bond and aromatic rings of EBT, breaking the molecule into small, non‑toxic fragments.
Under natural sunlight, the composite achieves >95 % removal of 20 mg L⁻¹ EBT within 8 minutes, with a first‑order rate constant of 0.38 min⁻¹ – substantially higher than undoped Ag₃PO₄ or ZnS alone.
Key Benefits for Real‑World Applications
- Speed: Fast kinetics reduce reactor volume and operating cost.
- Reusability: The catalyst retains >90 % activity after five cycles, thanks to La‑induced resistance to Ag⁰ formation.
- Scalability: Simple hydrothermal synthesis using inexpensive precursors makes large‑scale production feasible.
- Environmental safety: No harmful by‑products are detected; final water meets WHO standards for coloration and toxicity.
Practical Guidance for Implementation
Reactor Setup
1. Use a flat‑plate photoreactor to maximize solar exposure.
2. Maintain catalyst loading at 0.5 g L⁻¹ for optimal light penetration.
3. Adjust pH to 7–8; the catalyst works best under neutral conditions.
Operational Tips
• Pre‑mix the catalyst with wastewater for 5 minutes to ensure uniform dispersion.
• Periodically stir the solution to avoid sedimentation of ZnS QDs.
• After treatment, separate the catalyst by simple filtration or magnetic assistance if Fe‑doped variants are used.
Future Outlook
The success of La‑doped Ag₃PO₄/ZnS opens doors for:
- Designing other rare‑earth‑doped metal phosphates for diverse dyes.
- Hybridizing with conductive supports (e.g., graphene) to further boost electron mobility.
- Integrating the photocatalyst into pilot‑scale solar water‑treatment plants.
With continued research, sunlight‑driven photocatalysis could become the cornerstone of green textile wastewater management.
Conclusion
By combining La‑doping with ZnS quantum‑dot decoration, Ag₃PO₄ transforms into a powerhouse capable of rapidly mineralizing Eriochrome Black T under everyday sunlight. The approach delivers high degradation speed, excellent stability, and easy scalability—key ingredients for real‑world adoption. Embracing this technology can help industries meet stricter discharge regulations while moving toward a circular, low‑carbon future.

Comments are closed, but trackbacks and pingbacks are open.