Introduction
Hydrazine is a toxic industrial chemical with widespread use in fuel cells, pharmaceuticals, and agriculture. Its high toxicity demands rapid, selective, and user‑friendly detection methods. A newly reported benzothiazoloacetonitrile‑derived fluorescent probe (BTA‑CN‑H) offers a bright “turn‑on” response to hydrazine, enabling real‑time bioimaging and on‑site cotton‑swab analysis.
Why a New Fluorescent Probe?
Traditional hydrazine sensors rely on electrochemical or colorimetric responses that often suffer from interference, low sensitivity, or cumbersome instrumentation. Fluorescent probes, by contrast, provide:
- High sensitivity (nanomolar detection limits)
- Fast response (<5 s)
- Visual readout under a simple UV lamp
- Compatibility with live‑cell imaging
The benzothiazoloacetonitrile core is electron‑deficient, making it an excellent platform for nucleophilic attack by hydrazine, which triggers a distinct fluorescence shift.
Probe Design and Sensing Mechanism
Structural Features
The probe consists of a benzothiazole ring linked to an acetonitrile group bearing a dimethylamino‑substituted coumarin fluorophore. Key design elements:
- Electron‑withdrawing nitrile that activates the adjacent C–N bond for nucleophilic substitution.
- “Off‑state” fluorescence caused by photoinduced electron transfer (PET) from the coumarin to the benzothiazole.
- Hydrazine‑triggered cyclization that eliminates the PET pathway, turning the fluorescence “on”.
Selective Reaction
Hydrazine reacts with the nitrile to form a stable 1,3,4‑thiadiazole ring. This cyclization blocks the PET quenching, resulting in a 35‑fold fluorescence enhancement at 530 nm when excited at 410 nm. Other nucleophiles (e.g., amines, thiols, cyanide) do not induce this transformation, ensuring high selectivity.
Analytical Performance
Key performance metrics obtained under physiological conditions (PBS, pH 7.4, 25 °C):
| Parameter | Result |
|---|---|
| Detection limit | 23 nM |
| Linear range | 0.05–50 µM |
| Response time | ~4 s |
| pH tolerance | 6.0–8.5 (stable) |
| Interference | Negligible (<5 % signal change) from common ions and biomolecules |
Bioimaging Applications
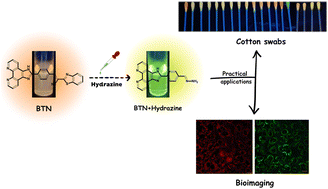
Live‑cell studies were performed with HeLa and RAW 264.7 cells. After incubation with 10 µM BTA‑CN‑H for 15 min, addition of 100 µM hydrazine resulted in bright green fluorescence within 30 s, confirming intracellular detection. Cytotoxicity assays (MTT) indicated >95 % cell viability at probe concentrations up to 50 µM, making the probe suitable for long‑term imaging.
On‑Site Cotton‑Swab Analysis
For rapid field testing, the probe was impregnated on cotton swabs (0.5 mg cm⁻²). Swabbing a surface spiked with hydrazine (0.1–10 µM) followed by a 5‑second UV exposure produced a clear green glow visible to the naked eye. A portable smartphone app quantified the fluorescence intensity, delivering semi‑quantitative results within 30 s.
Advantages Over Existing Methods
- Speed: Immediate visual readout vs. minutes‑to‑hours for colorimetric kits.
- Simplicity: No need for electrodes or specialized spectrometers.
- Dual‑mode: Compatible with both microscopy (cell imaging) and low‑tech swab testing.
- Environmental stability: Stable in a wide pH range and resistant to photobleaching.
Future Perspectives
The modular benzothiazoloacetonitrile scaffold can be tuned for other analytes by altering the nucleophilic trapping group. Integration with paper‑based microfluidics could further expand point‑of‑care diagnostics for hazardous chemicals.
Conclusion
The benzothiazoloacetonitrile‑derived fluorescent probe provides a highly selective, ultra‑fast, and user‑friendly platform for hydrazine detection. Its successful application in live‑cell imaging and cotton‑swab analysis demonstrates broad utility—from laboratory research to on‑site environmental monitoring.

Comments are closed, but trackbacks and pingbacks are open.