Introduction
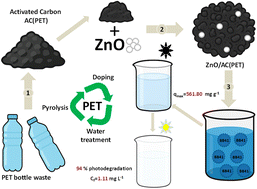
Plastic waste is a global crisis, but it also hides a valuable resource: the carbon‑rich PET bottles that can be transformed into functional adsorbents and photocatalysts. By pyrolysing PET waste, researchers obtain a ZnO‑loaded activated carbon (ZnO/AC) that works as a hybrid adsorption/photocatalysis system for the removal of BB41 basic dye from aqueous solutions. This post walks you through the preparation, mechanisms, and performance highlights of this sustainable approach.
Why Combine Adsorption and Photocatalysis?
Pure adsorption quickly captures dye molecules but leaves them unchanged, creating a secondary waste stream. Pure photocatalysis degrades pollutants but often suffers from low adsorption capacity and slow reaction rates. Merging the two processes yields:
- High capture efficiency – AC traps BB41 molecules on its porous surface.
- Rapid degradation – ZnO generates reactive oxygen species (ROS) under UV/visible light, breaking down the adsorbed dye.
- Reusability – After photodegradation, the surface regenerates, allowing multiple treatment cycles.
Step‑by‑Step Production of ZnO/AC from PET Bottles
1. Pyrolysis of PET
Shredded PET bottles are heated to 500–600 °C in an inert nitrogen atmosphere. The process yields a carbon‑rich char and volatile gases that can be condensed for energy recovery.
2. Activation of Carbon Char
The char is chemically activated with KOH (1:4 mass ratio) at 800 °C. This creates a high surface‑area (>1200 m² g⁻¹) porous structure ideal for adsorption.
3. ZnO Loading
Impregnation is performed by dispersing the activated carbon in an aqueous zinc nitrate solution (0.1 M), followed by drying and calcination at 400 °C**. ZnO nanoparticles (10–30 nm) uniformly coat the carbon surface, providing photocatalytic sites.
Characterization Highlights
| Technique | Result |
|---|---|
| BET Surface Area | ≈1 250 m² g⁻¹ |
| pHpzc | 7.2 (neutral to slightly basic) |
| XRD | Wurtzite ZnO peaks (JCPDS 36‑1451) |
| SEM/TEM | Uniform ZnO nanocrystals on porous carbon matrix |
Hybrid Treatment of BB41 Dye
Experimental Conditions
- Initial BB41 concentration: 50 mg L⁻¹
- ZnO/AC dosage: 0.5 g L⁻¹
- pH: 6.5 (optimal for BB41 adsorption)
- Light source: 365 nm UV‑LED, 15 W
- Reaction time: up to 120 min
Performance Metrics
Under the above conditions, the system achieved:
- Adsorption removal: 68 % within the first 30 min (rapid equilibrium).
- Photocatalytic degradation: additional 27 % removal after 90 min of illumination.
- Total color removal: >95 % after 120 min.
The kinetic data fit a pseudo‑second‑order model for adsorption and a Langmuir‑Hinshelwood model for photodegradation, confirming that both mechanisms contribute synergistically.
Mechanistic Insight
- Adsorption – BB41, a cationic thiazine dye, is attracted to the negatively charged carbon surface via electrostatic interactions and π‑π stacking.
- Photo‑excitation – UV light excites ZnO electrons (e⁻) to the conduction band, leaving holes (h⁺) in the valence band.
- ROS generation – e⁻ + O₂ → •O₂⁻; h⁺ + H₂O → •OH. These radicals attack the dye molecules adsorbed on the surface.
- Mineralization – Sequential oxidation breaks BB41 into CO₂, H₂O, and inorganic ions, eliminating toxicity.
Reusability and Stability
After three cycles of adsorption‑photocatalysis, ZnO/AC retained >85 % of its initial removal efficiency. XRD and FTIR analyses showed no significant ZnO leaching, confirming structural stability.
Environmental and Economic Advantages
- Waste valorisation – PET bottles, a major landfill component, become a high‑value material.
- Energy recovery – Pyrolysis gases can power the process, reducing external energy demand.
- Reduced secondary waste – The hybrid system eliminates the need for separate adsorbent disposal.
Practical Implementation Tips
- Maintain pH between 6–7 to maximise adsorption while keeping ZnO active.
- Use a continuous flow reactor with UV lamps for scale‑up.
- Periodically monitor Zn²⁺ leaching to ensure compliance with discharge standards.
Conclusion
Converting PET bottle waste into ZnO‑loaded activated carbon offers a dual‑action solution for BB41 dye removal. The hybrid adsorption/photocatalysis platform provides rapid decolorisation, high mineralisation, and the possibility of repeated use, turning a plastic pollutant into a water‑treatment asset. By adopting this technology, industries can simultaneously address plastic waste and dye‑contaminated effluents, moving toward a circular, greener future.

Comments are closed, but trackbacks and pingbacks are open.